Cervical cancer screening tests
- Pap Test/ Liquid Based Cytology
- HPV DNA Testing
What is cervical cancer screening?
Cervical cancer screening is used to find changes in the cells of the cervix that could lead to cancer. Screening includes cervical cytology (also called the Pap test or Pap smear), testing for human papillomavirus (HPV), or both. All women should have cervical cancer screening on a regular basis.
Cervical cancer caused by?
Most cases of cervical cancer are caused by infection with HPV. HPV is a virus. It enters cervical cells and can cause them to change. Some types of HPV have been linked to cervical cancer as well as to cancer of the vulva, vagina, penis, anus, mouth, and throat. Types of HPV that may cause cancer are known as “high-risk types.”
These and other types of HPV can be passed from person to person during sexual activity. HPV is very common—most people who are sexually active will get an HPV infection in their lifetime. HPV infection often causes no symptoms. Most people do not even know they are infected.
How is cervical cancer screening done?
Cervical cancer screening includes the Pap test/LBC, an HPV test, or both. Both tests use cells taken from the cervix. The screening process is simple and fast.
Patient is laid down on the examination table , vagina is exposed with a speculum Cells are removed from the cervix with a brush or other sampling instrument. The cells usually are put into a special liquid and sent to a laboratory for testing
- For a Pap test, the sample is examined to see if abnormal cells are present.
- For an HPV test, the sample is tested for the presence of the most common high-risk HPV types. Usually, the sample taken for the Pap test also can be used for the HPV test. Sometimes, two cell samples are taken. It depends on the type of Pap test that is used.
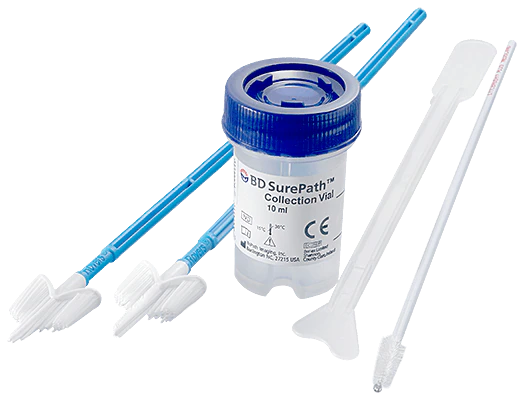
What is LBC/LIQUID BASED CYTOLOGY technology?
In LBC technology, a Cervex-Brush® (Rover’s Medical Devices BV, Oss, The Netherlands) or a combination of a plastic spatula and endocervical brush are used to collect the sample. Instead of spreading the cellular material removed from the cervix onto a glass slide, the samplers are rinsed, or the head of the broom is detached, into a vial of liquid transport medium creating a cell suspension. This cell suspension contains all of the cells removed from the cervix and will remain well preserved for several weeks at room temperature.
How often should cervical cancer screening be done?
The newest guidelines by american Obstetrics & Gynecology Society suggests the following
USPSTF Recommendation for Routine Cervical Cancer Screening
| Population | Recommendation | USPSTF Recommendation Grade |
|---|---|---|
| Aged less than 21 Years | No Screening | D |
| Aged 21 - 99 Years | Cytology alone every 3 years | A |
| Aged 30 - 65 Years | Any one of the following
|
A |
| Aged greater than 65 Years | No screening after adequate negative prior screening results | D |
| Hysterectomy with removal of the cervix | No Screening in individuals Who do not have a history of high-grade cervical precancerous lesions or cervical cancer | D |
Abbreviations: FDA, U.S. Food and Drug Administration; hrHPV, high-risk human papillomavirus testing.
These recommendations apply to individuals with a cervix who do not have any signs or symptoms of cervical cancer, regardless of their sexual history or HPV vaccination status. These recommendations do not apply to individuals who are at high risk of the disease, such as those who have previously received a diagnosis of a high-grade precancerous cervical lesion. These recommendations also do not apply to individuals with in utero exposure to diethylstilbestrol or those who have a compromised immune system (eg, individuals with human immunodeficiency virus)
Grade A denotes that "The USPSTF recommends the service. There is high certainty that the net benefit is substantial." A Grade D definition means that, "The USPSTF recommends against the service. There is moderate or high certainty that the service has no net benefit or that the harms outweigh the benefits." For more information on the USPSTF grades, see
Primary hrHPV testing is FDA approved for use starting at age 25 years, and ACOG, ASCCP, and SGO advise that primary hrHPV testing every 5 years can be considered as an alternative to cytology- only screening in average-risk patients aged 25-29 years
SAdequate negative prior screening test results are defined as three consecutive negative cytology results, two consecutive negative cotesting results, or two consecutive negative hrHPV test results within 10 years before stopping screening, with the most recent test occurring within the recommended screening interval for the test used (1, 5),
Data from Curry SJ, Krist AH, Owens DK, Barry MJ, Caughey AB, Davidson KW, et al. Screening for cervical cancer. U.S. Preventive Services Task Force recommendation statement. U.S. Preventive Services Task Force. JAMA 2018:320:674-86. Available at Retrieved April 12, 2021.
Another way to prevent Cervical cancer is by Vaccination against HPV. Gardasil vaccine is available at Aarya Women’s Hospital for prevention of HPV infection by type 6,11,16,18.
It is mandatory for all women who have been vaccinated to undergo routine screening as described above.